What Is TMS Therapy? A Complete Guide for Provo Residents
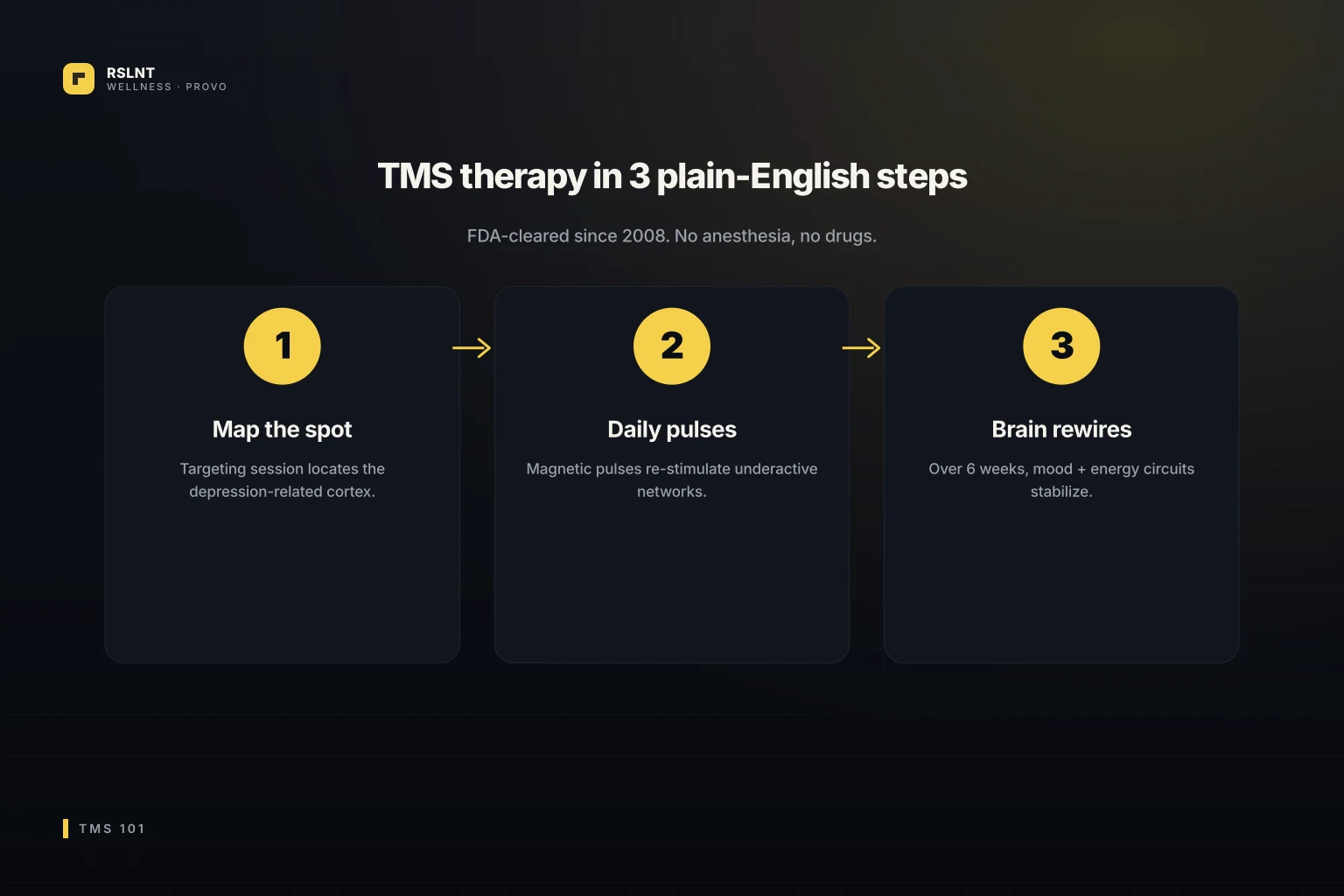
Table of contents
- What MagVenture TMS actually is, in plain language
- What a MagVenture session feels like
- Why we lead with MagVenture
- What treatment looks like start to finish
- Who TMS is and isn't for
- What the research shows for Provo, Utah, and beyond
- How we actually treat this at RSLNT
- Frequently asked questions
- The clearest answer comes from a 15-minute call
Your antidepressant didn't work. Maybe two didn't work. You're tired of side effects you didn't sign up for, and you're tired of feeling like a different version of yourself in a way that nobody warned you about.
Somebody mentioned TMS. You don't know what it is. You're sitting in front of your laptop trying to figure out whether it's real medicine or another wellness trend.
TMS therapy is real medicine. It stands for transcranial magnetic stimulation. At RSLNT Wellness, TMS means MagVenture: a MagVenture TMS Therapy System R30 with Express TMS, a Cool B70 butterfly coil on a Flow Arm, a MagPro R30 stimulator, and a MagVenture motorized procedure chair. It is non-invasive, FDA-cleared care that uses pulsed magnetic fields to activate underactive parts of the brain associated with depression, OCD, and anxiety-related symptoms that occur with major depression. For Provo and Utah County residents, treatment is available at RSLNT Wellness with sessions about 19 minutes each, five days a week for six weeks.
What MagVenture TMS actually is, in plain language
A MagVenture TMS setup is not just a generic chair. The patient sits in a MagVenture motorized procedure chair while a trained TMS technician positions the Cool B70 butterfly coil against a mapped treatment location on the scalp. The coil is held steady by the Flow Arm and powered by the MagPro R30 stimulator.
The coil sends short magnetic pulses, similar in strength to an MRI machine, into a specific spot, the dorsolateral prefrontal cortex. That spot matters because depression researchers have shown for decades that this region is often underactive in patients with major depression. The pulses don't add a chemical. They don't sedate you. They don't cross any blood-brain barrier. They prompt the brain's own circuitry to fire, repeatedly, until the underactive region wakes up.
Repeat that over weeks and the brain's neuroplasticity, the same mechanism that lets you learn a new language or recover from a stroke, builds new connections in the parts of the brain that handle mood and motivation.
It sounds like science fiction. It's not. It's standard psychiatric care now, and MagVenture is the platform RSLNT chose for that care.
What a MagVenture session feels like
Most patients describe the sensation as a woodpecker tapping on the side of a hard hat. Not painful. Slightly uncomfortable in the first few sessions while your scalp adjusts.
You sit in the MagVenture motorized procedure chair. The technician positions the Cool B70 coil with the Flow Arm. The pulses run in short bursts for around 19 minutes, depending on the protocol your clinician orders. You can read. You can scroll your phone. You can text your spouse. Some patients close their eyes and rest.
When the timer ends, you stand up, walk out, and drive yourself home. There's no anesthesia, no needles, no recovery time, and no driving restriction.
Why we lead with MagVenture
RSLNT chose MagVenture because the equipment matters to the patient experience. Our setup uses the MagVenture TMS Therapy System R30 with Express TMS, the Cool B70 butterfly coil, the MagPro R30 stimulator, and MagVenture's motorized procedure chair. That is the actual chair and device platform patients see in our Provo clinic.
The FDA history is important, but it needs to be said precisely. Tonica/MagVenture had an FDA 510(k) clearance for the MagPro R30 magnetic stimulator in 2006. That earlier clearance was for peripheral nerve stimulation for diagnostic purposes, not depression treatment. The depression-specific rTMS classification came through a different manufacturer's 2008 de novo/510(k) pathway. MagVita/MagVenture then received FDA clearance for major depressive disorder in 2015 and has continued expanding its cleared TMS indications.
So if you hear that MagVenture came first, that is true for MagVenture's magnetic-stimulation device lineage. If you hear that the first depression-specific FDA pathway came later through another TMS system, that is also true. What matters for RSLNT patients is this: we use MagVenture now, and MagVenture's current FDA-cleared TMS indications include major depressive disorder, MDD with comorbid anxiety symptoms in adults, adjunct treatment for OCD, and adjunct treatment for adolescent MDD in specific patient groups.
The American Psychiatric Association lists TMS as a recommended treatment for treatment-resistant major depressive disorder, with strong evidence supporting its use after two failed antidepressant trials.
What treatment looks like start to finish
Standard treatment is 36 sessions over about 6 weeks.
- Day 1: Motor threshold mapping. The clinician finds the exact spot on your skull above your motor cortex by giving small pulses until your thumb twitches. This calibrates the device to your specific brain. Around 45 minutes total.
- Days 2 through 20: Daily 19-minute sessions, Monday through Friday for 4 weeks.
- Weeks 5 and 6: Tapered sessions, going from daily to less frequent over two weeks.
Some patients add a maintenance session every 6 to 12 months once they respond.
Who TMS is and isn't for
Strong fits:
- Patients with major depressive disorder who haven't responded to two or more antidepressants
- Patients who can't tolerate antidepressant side effects
- Patients with OCD who haven't responded to SSRIs alone
- Patients in recovery from substance use who want a drug-free option
- Veterans on heavy medication stacks
Not a fit:
- Patients with metal implants in or near the head, including aneurysm clips, cochlear implants, or deep-brain stimulators (a pacemaker doesn't disqualify you, but disclosure matters)
- Patients with seizure disorders or active substance withdrawal
- Children under 18 (off-label clinical trials only)
- Patients who haven't yet tried a first-line antidepressant or therapy (we'd usually start there)
We screen for all of this in the first consultation.
What the research shows for Provo, Utah, and beyond
The largest meta-analyses on TMS for depression, including data from the National Institute of Mental Health and Stanford-led studies, show response rates of 50 to 65 percent and remission rates of 30 to 40 percent in patients with treatment-resistant depression. Real-world clinic numbers from established TMS centers are often slightly higher, in the 60 to 65 percent response range.
For comparison, antidepressant response rates after two prior failures drop to roughly 14 to 25 percent, per the STAR*D trial. For Provo and Utah County patients who have already tried two or three medications, TMS gives meaningfully better odds.
How we actually treat this at RSLNT
At RSLNT Wellness, our Provo clinic is built around MagVenture TMS technology and a complete care model with all three pillars under one roof.
Counseling that pairs with TMS. Patients who do therapy alongside TMS often see more durable results. Our clinicians use cognitive behavioral therapy and acceptance and commitment therapy. We schedule therapy on the same day as TMS when it fits.
Medication management that coordinates with whatever you're already taking. SSRIs like sertraline and escitalopram, SNRIs like venlafaxine, bupropion when motivation is the issue. We don't push pills. We don't withhold them either.
TMS therapy with full insurance verification before you start. We handle the prior authorization with SelectHealth, Regence, Cigna, Aetna, United, Medicare, TRICARE, and VA Community Care. Most patients pay $0 to $1,800 out of pocket after coverage. We tell you the exact number before session one.
Frequently asked questions
Is TMS the same thing as ECT?
No. ECT (electroconvulsive therapy) is a different treatment that uses electrical currents under anesthesia to induce a brief seizure. ECT is highly effective for severe, life-threatening depression, but it requires anesthesia and has cognitive side effects. TMS uses magnetic fields, no anesthesia, no seizure, and no cognitive side effects.
How fast does TMS work?
Some patients feel a small lift in week 2. The bigger shift typically lands between weeks 3 and 5. By the end of the six-week course, most patients see a meaningful drop in their depression scores.
What if it doesn't work?
About 35 to 40 percent of patients don't fully respond to a first TMS course. We have several next moves: extending the course, adjusting the target site, switching to an accelerated protocol like SAINT, returning to a different medication, or referring for ECT in severe cases. We don't drop you when one path doesn't work.
The clearest answer comes from a 15-minute call
If you're a Provo or Utah County resident considering TMS, the next move is a free assessment.
Schedule a free 15-minute consult. We'll talk about your history, what you've tried, and whether TMS makes sense for you. We'll verify your insurance and tell you the exact out-of-pocket cost before you commit.
I'm not a therapist or a doctor. I'm someone who went from suicidal ideation, major depressive disorder, and crippling anxiety to clarity of mind. I feel like I got my life back. RSLNT Wellness is the place that helped me get there. If you're struggling, you don't have to figure this out alone.

Frequently Asked Questions
Is TMS the same thing as ECT?
How fast does TMS work?
What if it doesn't work?
Sources & Further Reading
Every clinical claim in this article is backed by a public, peer-reviewed, or government source. We do not cite anything we cannot link to.
- [1]Transcranial magnetic stimulation - Mayo ClinicMayo ClinicBacks: TMS does not require anesthesia or sedation, and patients can return to normal activities after treatment.
- [2]FDA permits marketing of transcranial magnetic stimulation for treatment of obsessive compulsive disorderU.S. Food and Drug Administration · 2018Backs: The FDA permitted marketing of TMS for obsessive compulsive disorder in 2018.
- [3]TMS (Transcranial Magnetic Stimulation): What It IsCleveland ClinicBacks: Metal implants in or near the head may make TMS unsafe or unsuitable.
- [4]510(k) Premarket Notification K061645U.S. Food and Drug Administration · 2006Backs: FDA cleared Tonica/MagVenture MagPro R30 in 2006 for peripheral nerve stimulation for diagnostic purposes.
- [5]510(k) Premarket Notification K150641U.S. Food and Drug Administration · 2015Backs: FDA cleared MagVita TMS Therapy System for major depressive disorder in 2015.
- [6]FDA clearancesMagVenture · 2026Backs: MagVenture lists FDA-cleared indications for MDD, MDD with comorbid anxiety symptoms, OCD adjunct treatment, and other MagVenture TMS system clearances.
- [7]510(k) Premarket Notification K251119U.S. Food and Drug Administration · 2025Backs: FDA records list MagVenture TMS Therapy indication language for depressive episodes and decreasing anxiety symptoms in adult MDD patients with comorbid anxiety symptoms.
Ready to feel like yourself again?
Schedule a free consultation to see if TMS therapy is right for you.
Book Your Consultation